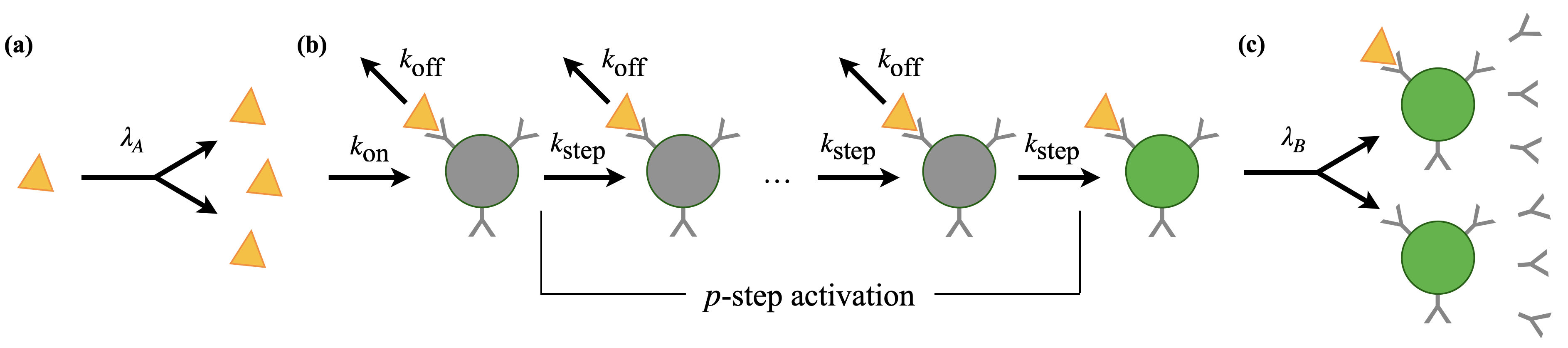

Abstract: The immune response to an acute primary infection is a coupled process of antigen proliferation, molecular recognition by naive B cells, and their subsequent proliferation and antibody shedding. This process contains a fundamental problem: the recognition of an exponentially time-dependent antigen signal. Here we show that B cells can efficiently recognise new antigens by a tuned kinetic proofreading mechanism, where the molecular recognition machinery is adapted to the complexity of the immune repertoire. This process produces potent, specific and fast recognition of antigens, maintaining a spectrum of genetically distinct B cell lineages as input for affinity maturation. We show that the proliferation-recognition dynamics of a primary infection is a generalised Luria-Delbrück process, akin to the dynamics of the classic fluctuation experiment. This map establishes a link between signal recognition dynamics and evolution. We derive the resulting statistics of the activated immune repertoire: antigen binding affinity, expected size, and frequency of active B cell clones are related by power laws. Their exponents define the class of generalised Luria-Delbrück processes; they depend on the antigen and B cell proliferation rate, the number of proofreading steps, and the lineage density of the naive repertoire. Empirical data of mouse immune repertoires are found to be consistent with activation involving at least three proofreading steps. Our model predicts key clinical characteristics of acute infections, including the emergence of elite neutralisers and the effects of immune ageing. More broadly, our results establish acute infections as a new probe into the global architecture and functional principles of immune repertoires.
Abstract: Global strategies to contain a pandemic, such as social distancing and protective measures, are designed to reduce the overall transmission rate between individuals. Despite such measures, essential institutions, including hospitals, schools, and food producing plants, remain focal points of local outbreaks. Here we develop a model for the stochastic outbreak dynamics in such local communities. We derive analytical expressions for the probability of containment of the outbreak, which is complementary to the probability of seeding a deterministically growing epidemic. This probability depends on the statistics of the intra-community contact network and the initial conditions, in particular, on the contact degree of patient zero. Based on this model, we suggest surveillance protocols by which individuals are tested proportionally to their degree in the contact network. We characterize the efficacy of contact-based protocols as a function of the epidemiological and the contact network parameters, and show numerically that such protocols outperform random testing.